Student article – Regulation of Integrin Alpha 6 in relation to myelination. Rachel Gump
Regulation of Integrin Alpha 6 in Relation to Myelination – An Exploration of Peripheral Nervous System Repair

Rachel Gump. Biology/Neuroscience Age: 18 years
Key Words: Integrin Alpha 6, Schwann cell, Genotype, Peripheral Nervous System, Myelin
Abstract
The aim of my project was to study the impact of specific genotypes on the differentiation of Schwann cells1, and therefore the subsequent effect on myelination, post severe nerve injury2,3. This involved the dissection of Sciatic nerves from mice, with genetic combinations to result in the genotype I (-Fl/Fl-). These mice thus did not produce the correct amount of Integrin Alpha 6 to repair their nerves effectively after an injury. The Polymerase Chain Reaction (PCR) confirmed this. I was also a part of exploring options for cancer treatments. The results gained from the nerve research, that is the quantifying of the amounts of protein in relation to the myelin present, enlightened further the role of protein pathways in differentiation of specialised cells through the body. Applying my research, medicines are now being trialled to see their effect on stunting the growth of Schwannoma cell tumours by blocking the Hippo pathway.
Background
My research began as a placement through the Nuffield Foundation at Plymouth University to gain work experience. However, this quickly snowballed into key findings in the neuroscience community that allowed me to explore the field deeper.
To start, I had to carry out some scholarly research into the origins of my project4,5. This involved reading previous reports by neuroscientists, including Glenn & Talbot1, Parkinson2,3, and Xu6. Surprisingly, most of my reading led me to Santiago Ramon y Cajal (1852 – 1934) who is widely regarded as the father of neuroscience7. Artist turned neuroscientist, he published many works describing his theory on the function of the nervous system as a means of several working parts to coalesce into one entity, and drew many structures in the brain, including Purkinje cells named after their discoverer, Czech anatomist Jan Evangelista Purkyně in 1839 (Figure 1). Cajal, who won the Nobel prize in 19068, became a major inspiration for my research, and allowed me to have a greater appreciation for how far modern science has come, and how much can change in such a short period of time. He was the first to describe the Purkinje cells in the brain named after their discoverer, Czech anatomist Jan Evangelista Purkyně in 1839 (Figure 1).
Figure 7 Drawing of Purkinje cells
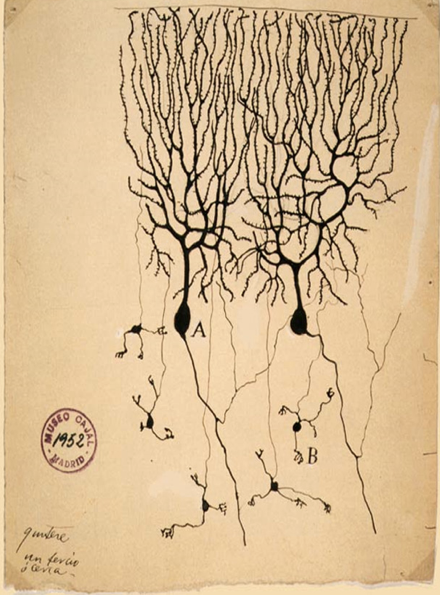
(A) and granule cells (B) from pigeon cerebellum by Santiago Ramón y Cajal, 1899. Instituto Santiago Ramón y Cajal, Madrid, Spain.
Public domain – https://commons.wikimedia.org/wiki/File:PurkinjeCell.jpg. This is done with ink on paper, using microscope images. This shows the intense way that nerves can lace together to form a network whilst still being individual. This is done with ink on paper, using microscope images.
Methods
PCR: During the course of my placement, a primary technique used was PCR. This is used with DNA samples to test for the presence of proteins, namely focusing on the absence of TAZ9. PCR allows the researcher to easily distinguish the genotype of the different DNA samples. My purpose of the genotyping was to find the correct nerve sample to view the myelination levels and compare to the control (Figure 2).
Staining and Imaging: After sections have been cut and placed on the slides, ensuring the slide is positively charged, it is now time to stain the nerve sample. This process of staining is done by attaching layers of antibodies to the cells, in an effort to illuminate the different areas of the cell needed to document. Essential to staining being successful, the slide must first be permeabilized using a solution of BSA and Triton.
Figure 8. PCR showing necessary genotype
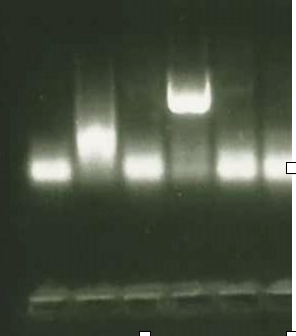
Well 4 shows that the sample is a TAZ knockout genotype, and well 5 has both copies of the TAZ gene present. These bands are illuminated by UV light on agarose gel.
This allows the antibodies to enter the cell, across the cell membrane, and attach to the correct sites. Antibodies must be picked carefully for staining, and the appropriate layering system must be used to ensure accurate and clear images. These theory-based questions should be answered before attempting to stain, as it will impact how staining is done and in what order. For my first staining, I used a two layer system with Hoescht dye and integrin alpha 6 (Figure 3).
Figure 9. Nerve with Integrin Alpha 6 Illuminated against Hoescht Dye.
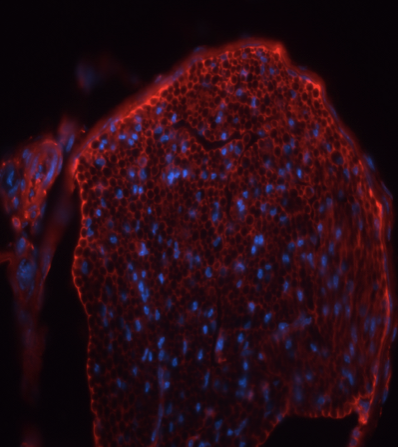
This is taken using the confocal microscope. A series of images are overlaid to get this final result. The honeycomb pattern shows the level of myelination present around the nerve axons.
Imaging: I used a variety of microscopes to ensure the images I gained were of a high quality and magnification, to fully quantify and compare the myelination present. The images below are taken from the Confocal microscope, which uses laser excitation to contrast the colors on the proteins.
Results
The positive results from the PCR allowed me to unequivocally know that the nerve I was staining and examining was indeed possessing the correct experimental genotype. Using an optical microscope to check that the stain and section of nerve had taken was also an important step before imaging, and must also be a positive result. Imaging allowed the conclusion to be reached that the protein levels are indeed significant to the myelination of the nerve. These results were also scrutinised for error, for example, a contamination in the PCR step could alter the results and skew the conclusions of the investigation (Figure 4).
Figure 10. Water Contamination PCR (lower left well)
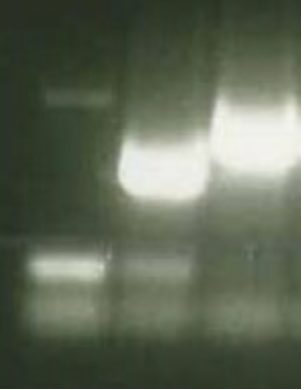
The slight illumination here shows that there has been cross contamination in the control, and that these results may not be fully accurate. Therefore another PCR must be run.
A contamination is seen when the band on a control sample is illuminated, pointing to human error in setting up the apparatus.
Conclusions
To conclude, my project has taken small steps toward helping a much bigger picture: to figure out exactly which proteins affect the peripheral nervous system’s repair, and could be involved in pain10. More specifically, I was trying to determine the difference in levels of myelination between nerves with contrasting genotypes of Fl/fl CRE+ or fl/fl CRE-. Initial, significant results culminated in the final imaging of the nerves with a confocal and/or Zeiss microscope, capable of high quality photography. These images captured are then used in a number of fascinating ways, such as counting the tissues present, observing and comparing differing samples, and further photography with a multitude of different apparatuses.
Acknowledgements
I would like to sincerely thank everyone involved in my project, from the very beginning. Mr. Andrew Petherick helped me to gain the research position in the first place, along with Dr. Peter Maunder and Mr. Robert Taylor, both fantastic teachers at Callington Community College. I would also like to acknowledge Mrs. Suzanne Davis-Butler, and Mr. Matthew Hortop and Mrs. Simone Bremnar who all helped me to coordinate and plan my various displays for competing with my project. Mrs. Cath Roberts and Mr. Alan Yendell deserve to be thanked as well, due to their diligent work inside our Sixth Form and in making sure I have always had exactly what I need to succeed. Finally, I would like to acknowledge Professor David Parkinson and Rachel Delourme, and all those at Nuffield Foundation and Peninsula Medical School who helped me extensively with my research placement, and beyond into competitions and volunteering opportunities that have truly been once in a lifetime. I assure you all that your generosity will never be forgotten, and that I will do my utmost to continue to make Callington proud!
Bibliography
1. Glenn, T. D., & Talbot, W. S. (2013). Signals Regulating Myelination in Peripheral Nerves and the Schwann Cell Response to Injury. SciVerse ScienceDirect, 1041-1048.
2. Parkinson, D. E. (2013). Plastic Fantastic: Schwann Cells and Repair of the Peripheral Nervous System. Stem Cell Translational Medicine, 1-6.
3. Parkinson, D. E. (2017). Sox 2 expression in Schwann Cells Inhibits Myelination in vivo and induces influx of Macrophages to the Nerve. The Company of Biologists, 1-12.
4. Sotelo, C. (2003). Viewing the Brain through the Master Hand of Cajal. Perspectives, 1-4.
5. Swanson, L and Newman, E. (2017). The Beautiful Brain: The Drawings of Santiago Ramon y Cajal. Abrams Books, USA.
6. Xu, N. e. (2016). Inhibition of YAP/TAZ Activity in Spinal Cord Suppresses Neuropathic Pain. Journal of Neuroscience 36, 10128 -0 10140.
7. Lobato, R. (2007). Historical Vignette of Cajal’s Work ‘Degeneration and Regeneration of the Nervous System’ with a reflection of the author. Neurocirugia, 456-468.
8.Nobel Prize Winners. (2014). Nobel Prize in Physiology or Medicine 1906. Retrieved September 3, 2017, from https://www.nobelprize.org/nobel_prizes/medicine/laureates/1906/cajal-bio.html.
9. Poitelon, Y. E. (2016). YAP and TAZ control peripheral myelination and the expression of laminin receptors in Schwann cells. Nature Neuroscience.
Reviewers comment:
This is not only exceptional molecular biology, but a beautifully written report. My favourite aspect was the acknowledgement that her water control would need to be run again. Dr Keith Hart, Cardiff University.
1 Comment
| Contents Issue 2
[…] Regulation of Integrin Alpha 6 in Relation to Myelination – An Exploration of Peripheral Nervo… […]